


Several options are available to achieve visual rehabilitation in patients with keratoconus to avoid or delay the need for lamellar and penetrating keratoplasty. They include spectacles, contact lenses, the implantation of intrastromal corneal ring segments (ICRSs), corneal surface ablation combined with CXL, and the implantation of phakic IOLs. Now, another exciting option is on the horizon: allograft corneal rings. This article explores the potential of these technologies.
AVOIDING THE PROBLEMS ASSOCIATED WITH RING SEGMENTS
Most ICRSs currently in use are made of PMMA. These segments are implanted in the eye to induce a geometric change in the central corneal curvature, thereby improving visual acuity. One drawback to implanting these devices is that it can be associated with serious complications, including corneal melting and extrusion in eyes with thin corneas (Figure 1). The latter can require urgent keratoplasty and lead to permanent vision loss. Other potential complications include segment migration, endothelial perforation, corneal haze, and misdirection of the segment into deeper or more superficial layers of the cornea. Further, implanting a foreign body into the cornea may result in a loss of corneal transparency and subepithelial stromal opacity in the long term.1,2
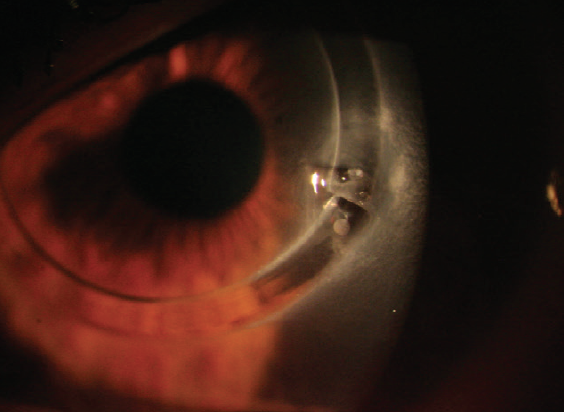
Figure 1. Extrusion of a biosynthetic (PMMA) ICRS.
Surgeons have turned their attention to the use of ICRSs made of allogenic corneal tissue as a potential means of improving the safety of this procedure. The use of allogenic tissue provides biocompatibility and improved integration into the cornea and avoids problems such as inflammation related to the insertion of a synthetic material into the cornea.3 Allogenic corneal tissue provides an unhindered passage of oxygen and nutrients to the cornea, ensuring stable corneal conditions and decreasing the risks of corneal necrosis and melt.4,5
ALLOGENIC TISSUE ALTERNATIVES
There are currently two allogenic tissue alternatives being studied.
No. 1: Corneal allogenic intrastromal ring segments. Jacob and colleagues first described the combination of allogenic ICRS implantation and CXL for the management of keratoconus.6 The allogenic ring segments are prepared from human donor corneal tissue and placed in the midperipheral stroma. (For more information, see A Novel Allogenic Donor Corneal Therapy.)
A Novel Allogenic Donor Corneal Therapy
CAIRS can be performed as a standalone procedure for keratoconus or in combination with CXL.
By Soosan Jacob, MS, FRCS, DNB

Corneal allogenic intrastromal ring segment (CAIRS) implantation, introduced in 2015, refers to the implantation of unprocessed or processed allogenic tissue for the treatment of keratoconus.1 Performed as a standalone procedure or in combination with CXL to strengthen the cornea, allogenic intrastromal corneal ring segments (ICRSs) are implanted at 50% corneal depth.
The use of donor corneal tissue avoids the possible complications associated with implanting synthetic ICRSs in the cornea. These include implant extrusion, intrusion, and migration; neovascularization; corneal melt and necrosis; and infection.
The allogenic ICRSs used in CAIRS are obtained from either processed donor tissue or a donor corneoscleral rim that has been deendothelialized and deepithelialized. The tissue is then cut into the desired shape and thickness with a double-bladed trephine (patent pending). Allogenic ICRSs are biocompatible, and they are therefore less likely to be extruded and to migrate compared with synthetic ICRSs. Integration of a shallower insertion plane (50% corneal depth vs 70% to 80% with synthetic implants) may also enhance the ability of an allogenic ICRS to reshape the cornea, meaning it can be used in a wider range of eyes with more severe corneal thinning and/or steepening.1
SURGICAL STEPS
A video of the procedure can be viewed at bit.ly/jacob0221. A femtosecond laser–dissected channel is created with two entry incisions located 180º apart; alternatively, a manual tunnel may be used. The allogenic tissue is pushed in gently from one side of the channel arc with a blunt rod and then pulled in from the other side with a reverse Sinskey hook. These movements are repeated with a second segment to form the second half-arc of the channel. The ends of the tissue are then trimmed and positioned within the channels. Single or double segments may be used.
Once the segments are in place, epithelium-off accelerated CXL or contact lens–assisted CXL may be performed if a two-stepped procedure is planned.
CONCLUSION
In my experience with CAIRS, patients achieved good refractive and topographic outcomes, regularization of the cornea, and centralization of the cone (Figure). Aberrations and regular and irregular astigmatism are also reduced. I have used this technique to treat all stages of keratoconus, from mild to advanced.
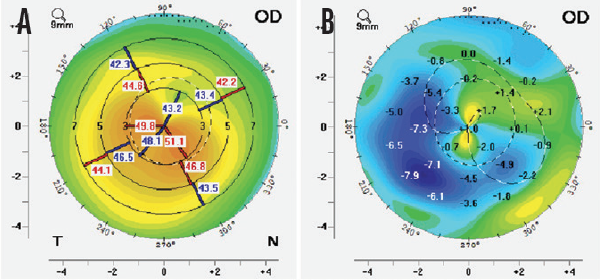
Figure. Preoperative (A) and difference (B) keratometric maps of a patient with keratoconus. Note the regularization of topography and centralization of the cone.
1. Jacob S, Patel SR et al. Corneal allogenic intrastromal ring segments (CAIRS) combined with corneal cross-linking for keratoconus. J Refract Surg. 2018;34(5):296-303.
No. 2: Sterile allograft rings. Lions VisionGift has developed sterile allograft rings and arcs that are packaged for ease of use. These KeraNatural products have a shelf life of 2 years, which offers greater convenience to patients and surgeons.
ISTANBUL PROTOCOL
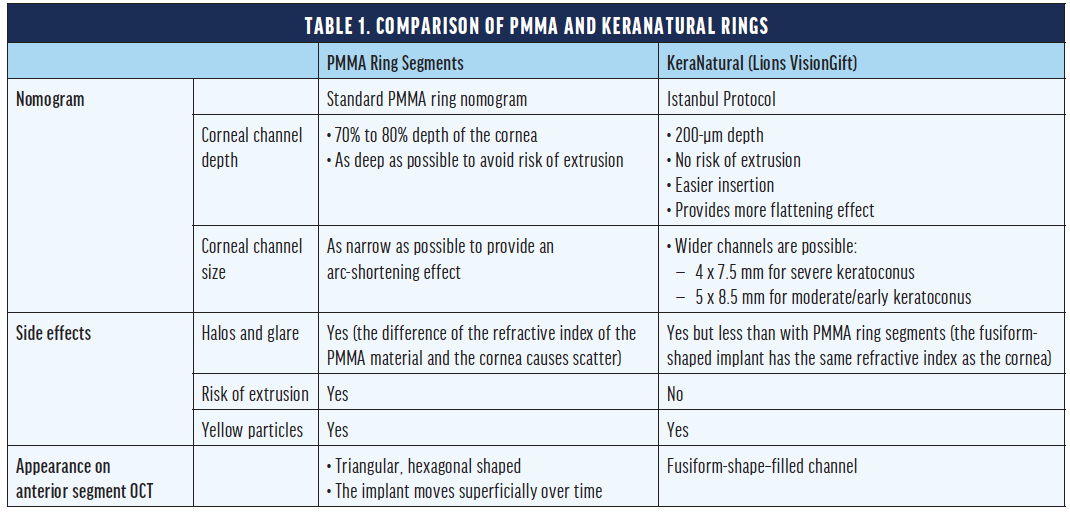
Current ICRS nomograms are formulated for rigid segments, but the biomechanical effects on the cornea of segments composed of PMMA and a (softer) allograft differ significantly in our experience. For this reason, we developed a special nomogram for the implantation of KeraNatural rings and arcs that we dubbed the Istanbul Protocol, and we reported our initial clinical results at the ESCRS 2020 Annual Meeting.7 When following this protocol, a wider and shallower corneal channel is used (see The Istanbul Protocol). The properties and differences of ICRS and allograft corneal rings are found in Table 1.
Istanbul Protocol Nomogram
- 180º arc, full-thickness corneal tissue (around 500 µm)
- 2 mm wide
- Channel diameters and depth
- Inner diameter: 4 mm
- Outer diameter: 7.5 mm
- Depth: 200 µm
- Incision cut energy: 1.3 µJ
- Ring energy: 1.3 µJ
- Incision
- Cut length: 1.5 mm
- Axis: steep
- Cones
- Asymmetric cone: single segment implantation
- Central cone: symmetric two-segment implantation
In a retrospective, nonrandomized, noncomparative, consecutive case series, we implanted KeraNatural ring segments using the Istanbul Protocol in 45 eyes of 32 patients with keratoconus. The corneal tunnels were created with a femtosecond laser at a depth of 200 µm and a diameter of 4 x 7.5 mm.
Pre- and postoperative evaluations are found in Table 2. There were statistically significant improvements in uncorrected distance visual acuity, corrected distance visual acuity, spherical equivalent, and mean keratometry. Statistically significant reductions in anterior maximum and mean elevations and posterior maximum and mean elevations were observed after the surgeries. We concluded that the implantation of an allograft corneal ring produced statistically significant changes and significant flattening in the anterior and posterior surfaces of the cornea.
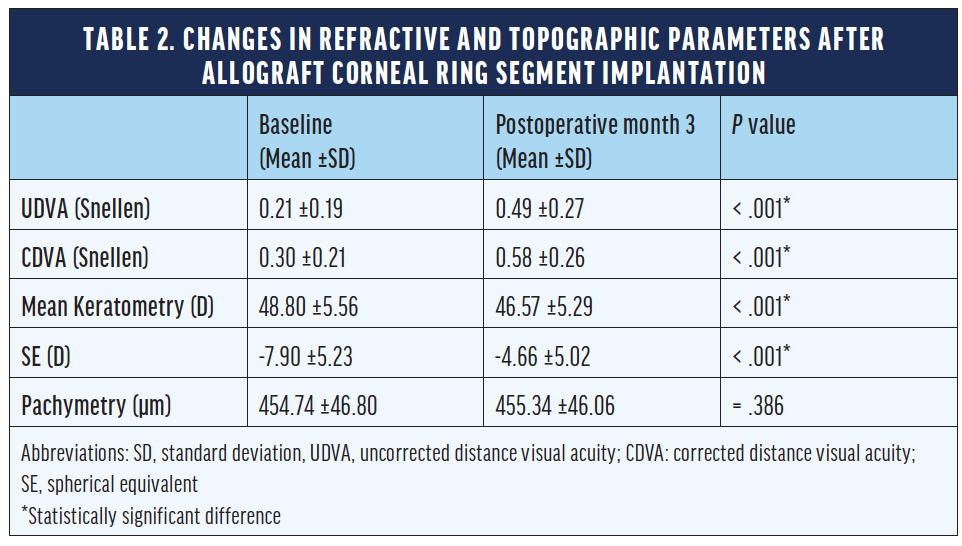
We also determined that the fusiform shape of the KeraNatural filled the corneal channel in a natural way. As a result, we anticipate more regular anterior and posterior corneal surfaces compared to preoperative measurements (Figure 2). Moreover, none of the patients in our series reported glare, halos, or light scatter—common visual phenomena after the implantation of biosynthetic ICRSs. The difference in photic phenomena may relate to the softness of the allogenic material, how it fills the corneal channel, and its biocompatibility.
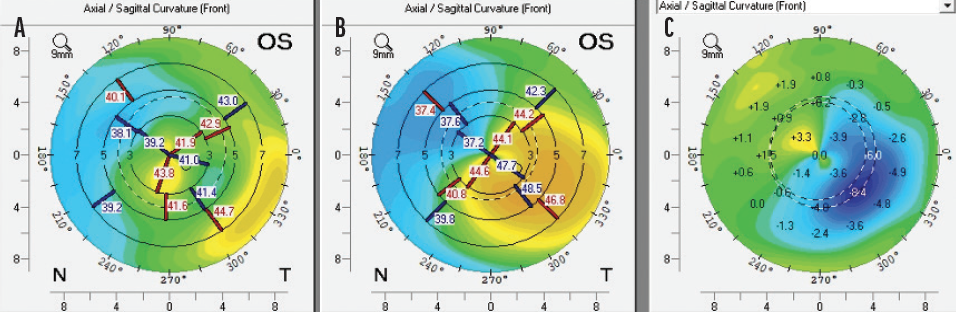
Figure 2. Postoperative (A), preoperative (B), and topographical difference maps (C) of one eye that received an allograft corneal ring.
CONCLUSION
We believe that the future of ICRS implantation may lie with devices composed of allogenic tissue. Our early research shows that this method of keratoconus treatment appears to offer greater safety and fewer side effects compared to use of biosynthetic implants. The pool of candidates may also be wider with KeraNatural because the risk of extrusion is lower with this material.
1. Giacomin NT, Mello GR, Medeiros CS, et al. Intracorneal ring segments implantation for corneal ectasia. J Refract Surg. 2016;32(12):829-839.
2. Sakellaris D, Balidis M, Gorou O, et al. Intracorneal ring segment implantation in the management of keratoconus: an evidence-based approach. Ophthalmology and Therapy. 2019;8(1):5-14.
3. Riau AK, Liu YC, Yam GH, Mehta JS. Stromal keratophakia: corneal inlay implantation. Prog Retin Eye Res. 2020;75:100780.
4. Kilic A, Burcu NT, Merve O, Muller D, Mrochen M. Excimer laser shaped allograft corneal inlays for presbyopia: initial clinical results of a pilot study. Clin Exp Ophthalmol. 2019;10:1000820.
5. Fenner Beau J, Yu-Chi L, Koh SK, et al. Mediators of corneal haze following implantation of presbyopic corneal inlays. Invest Ophthalmol Vis Sci. 2019;60(4): 868-876.
6. Jacob S, Patel SR, Agarwal A, Ramalingam A, Saijimol AI, Raj JM. Corneal allogenic intrastromal ring segments (CAIRS) combined with corneal cross-linking for keratoconus. J Refract Surg. 2018;34(5):296-303.
7. Hacıağaoğlu S, Tanrıverdi C, Özpınar A, Kılıç A. Allograft corneal ring for keratoconus management: Istanbul Protocole initial clinical results. Poster presented at the: 38th Congress of ESCRS Virtual Meeting 2020; October 2–4, 2020.



