
The market for multifocal IOLs is growing, and manufacturers continue launching new models year after year. The expectations of patients and surgeons, however, are advancing at a faster pace than the technological evolution in lens design. In an attempt to gain more market share, some manufacturers use confusing marketing messages and claim that their IOLs offer advantages over competitors’. In actuality, these claims are often based on minor changes in lens design. The Table summarizes the multifocal IOLs that
The Table summarizes the multifocal IOLs that are commercially available. These IOLs use refractive and diffractive principles, usually in combination, to achieve multifocality. IOL classification becomes more complex when it takes into account physical characteristics of a lens such as apodization, achromatic structure, and the number and shape of the rings (sinusoidal or echelette).
SIMPLE CLASSIFICATION SYSTEMS MAY BE CONFUSING
The introduction of extended depth of focus (EDOF) and monofocal-EDOF, also known as enhanced monofocal, IOLs adds another layer of complexity to lens classification systems. Using the defocus curves of IOLs rather than the dioptric additions to classify and compare multifocal IOL technologies may provide more useful information centered on patient performance.
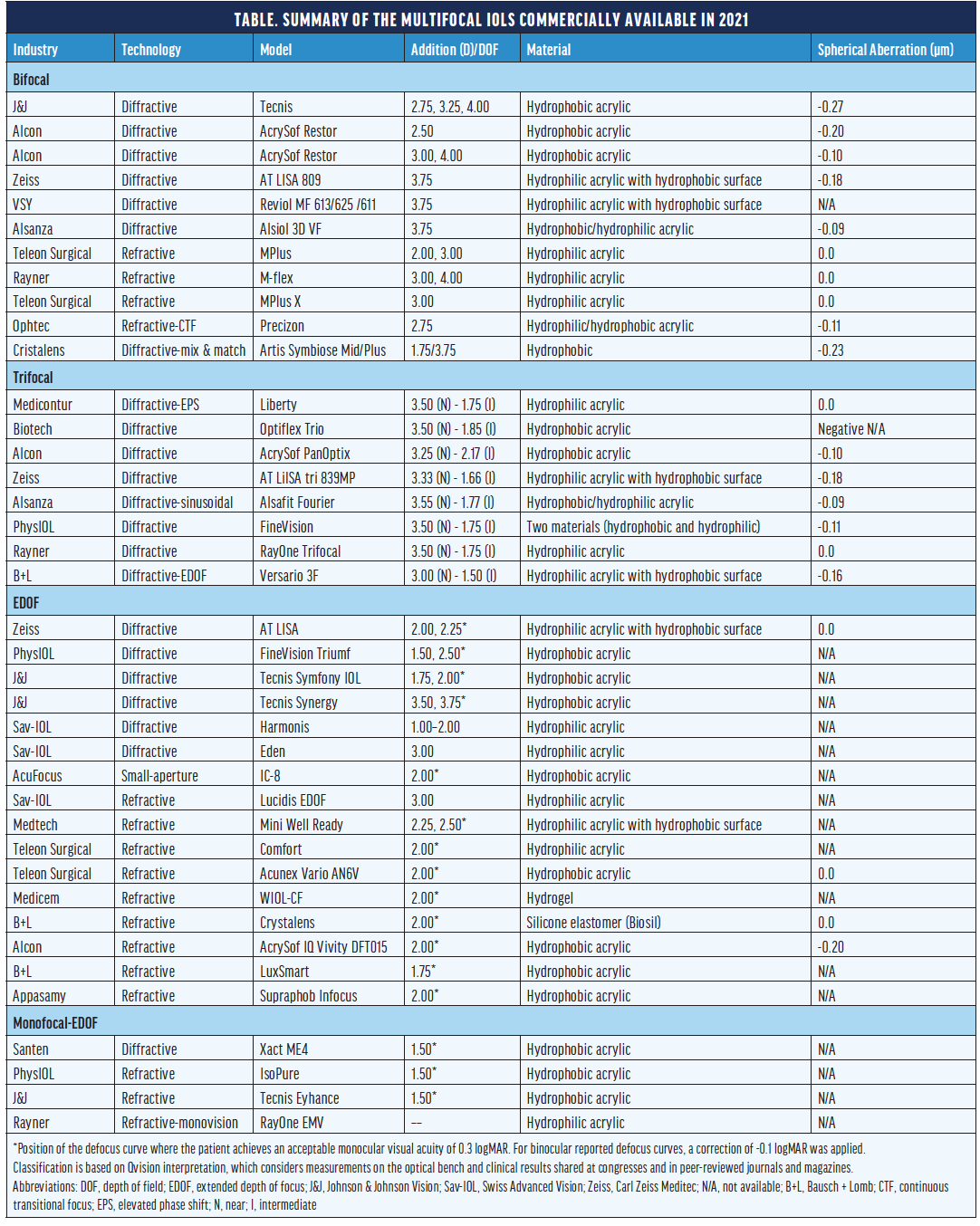
The American National Standards Institute (ANSI) describes depth of focus as “the dioptric power from the best peak of vision up to the defocus point for which the visual acuity is 0.2 logMAR.”1 The yellow line in Figure 1 represents the depth of focus (1.25 D) for one multifocal IOL (labeled as MIOL5). For EDOF and enhanced monofocal IOLs, the ANSI states that the lens must achieve an increase of 0.50 D in depth of focus compared to a control monofocal IOL. This information, however, is hard to find in scientific publications and presentations. Thus, we considered the dioptric power at the visual acuity cutoff (0.3 logMAR) to describe the performance of EDOF and monofocal-EDOF IOLs listed in the Table rather than the ANSI standard. Multifocal IOLs for which an addition at the IOL plane is provided by the manufacturer are categorized as bifocal or trifocal IOLs in the Table. Some manufacturers of EDOF IOLs report that an addition can be obtained from optical bench measurements. For those EDOF IOLs, the addition is shown without an asterisk in the Table. This exercise shows the flaw in simple classifications of bifocal, trifocal, EDOF, and monofocal-EDOF IOLs. The Artis Symbiose (Cristalens) provides an example. The Artis Symbiose Mid model of this IOL features a continuous transition from near to intermediate, whereas the Artis Symbiose Plus model features a continuous transition from intermediate to near. Should this IOL be classified as a bifocal, a trifocal, both, or neither? It is hard to tell with the simple classification system typically used today.
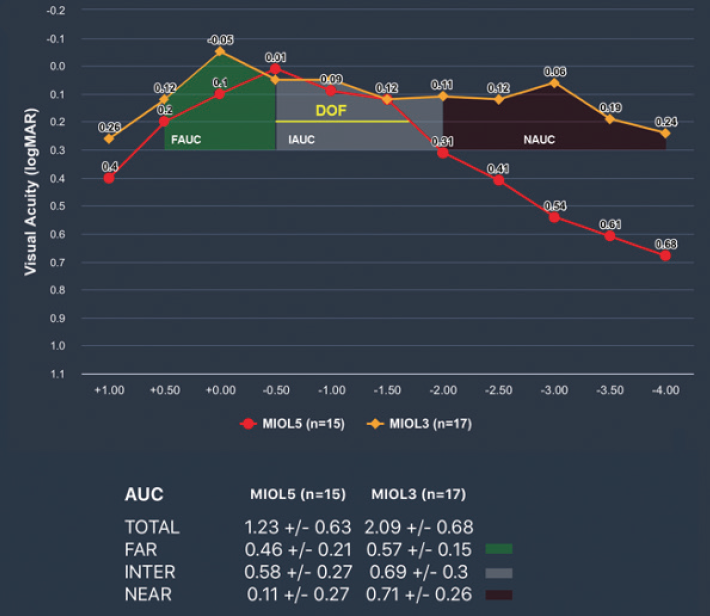
Figure 1. Defocus curve analysis conducted with the Multifocal Lens Analyzer 3.0. Comparison of two multifocal IOLs measured with visual acuity defocus curves.
Another example is the Tecnis Synergy (Johnson & Johnson Vision), which is classified as an EDOF IOL in the Table. This lens provides a more extended range of vision than all of the other EDOF IOLs.2 Should it be classified as an EDOF IOL or a bifocal? This will depend on future clinical results showing a monotonic decrease or nonmonotonic variation of visual acuity defocus curve.1
These are only two examples of the controversy that may result from the use of this simple classification system for multifocal IOLs.
AREA UNDER THE DEFOCUS CURVE: A BETTER METHOD
Considering the current difficulty surrounding the classification of multifocal IOLs, the Qvision Research+Evidence department* began to explore alternative methods of lens classification. We found that modifying classification to focus on standard clinical outcomes (eg, visual acuity at distance, intermediate, and near) is not the best option. This is because multifocal IOLs have differing dioptric additions in the optic, so the peak of intermediate and near vision is achieved at various distances with different lenses. In fact, the biometric parameters of different eyes can result in variations in near visual performance with the same multifocal IOL.3
Another approach we considered was to use the defocus point established by ANSI Z80.35-2018 for EDOF lenses to produce 0.2 logMAR visual acuity or the defocus point to produce 0.3 logMAR visual acuity, as noted in the Table. However, this classification system dismisses important information; an EDOF lens that provides 0.2 logMAR visual acuity at -2.00 D of defocus and better visual acuities from 0 to -1.50 D is different from a lens that provides continuous visual acuity of 0.2 logMAR along a defocus point from 0 to -2.00 D.
Use of the area under the defocus curve (AUC) solves the problem of the ANSI approach to lens classification. It considers not only the depth of focus but also the performance achieved along the depth of focus. Figure 1 shows how the defocus ranges.
An AUC above 0.3 logMAR was first reported in 2012 by Buckhurst et al4 and later by others.5,6 Despite the benefits of the AUC for comparing the performance of multifocal IOLs, use of the AUC in IOL classification systems has not been extended to clinical practice. This is likely due to the complexity of the calculation.
MAKING THE AUCS ACCESSIBLE TO PRACTITIONERS
To promote the use of the AUC as a new metric in lens classification, we developed the Multifocal Lens Analyzer (defocuscurve.com).7 To validate its use,8 we looked for evidence of contrast sensitivity defocus curves (CSDCs), the benefits compared to visual acuity defocus curves (VADCs) for detecting changes in optical quality,9,10 and the relationships between the AUC and biometric eye parameters.3,9,11 We also explored new metrics such as chromatic CSDCs.12
Figure 2 shows how CSDCs result in steeper defocus curves compared to the VADCs in Figure 1. This is because the same change in optical quality produces twice the effect in CSDC as in VADC.10
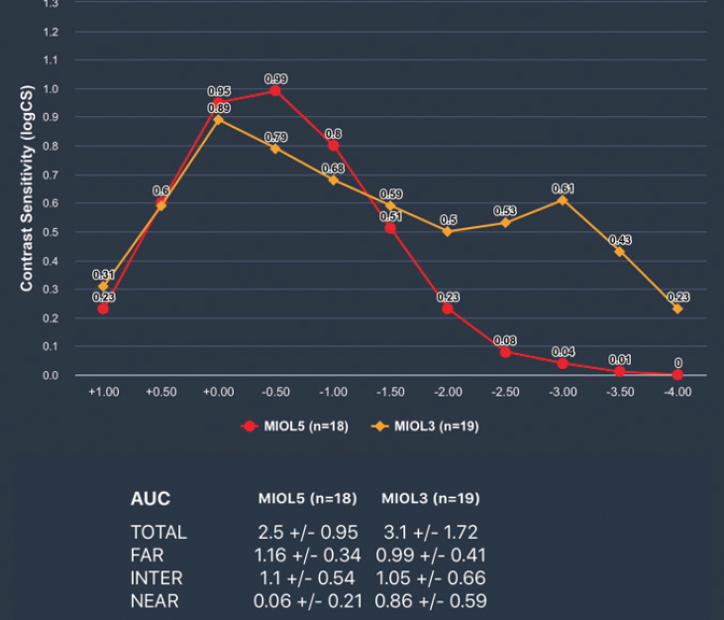
Figure 2. The same multifocal IOLs as in Figure 1, this time measured with contrast sensitivity defocus curves.
The Multifocal Lens Analyzer 3.0 automatically computes the AUC after each measurement. As a result, AUCs can be compared between multifocal IOLs in four possible regions:
- Total AUC (+1.00 to -4.00 D in 0.50 D steps);
- Distance AUC (+0.50 to -0.50 D);
- Intermediate AUC (-0.50 to -2.00 D); and
- Near AUC (-2.00 to -4.00 D). Use of the Multifocal Lens Analyzer
to classify multifocal IOLs based on their performance at total distance, intermediate, and near.
CONCLUSION
In the future, we hope that all practitioners can use a functional lens classification system based on AUCs. In the meantime, we have developed a standardized protocol so that users from around the world can share their results. This is available at www.defocuscurve.com/en/standard.
1. Akella SS, Juthani VV. Extended depth of focus intraocular lenses for presbyopia. Curr Opin Ophthalmol. 2018;29:318-322.
2. Rampat R, Gatinel D. Multifocal and extended depth-of-focus intraocular lenses in 2020. Ophthalmology. September 25, 2020. doi:10.1016/j.ophtha.2020.09.026
3. Fernández J, Rodríguez-Vallejo M, Martínez J, Tauste A, Piñero DP. Biometric factors associated with the visual performance of a high addition multifocal intraocular lens. Curr Eye Res. 2018;43:998-1005.
4. Buckhurst PJ, Wolffsohn JS, Naroo SA, et al. Multifocal intraocular lens differentiation using defocus curves. Invest Opthalmol Vis Sci. 2012;53:3920.
5. Wolffsohn JS, Jinabhai AN, Kingsnorth A, et al. Exploring the optimum step size for defocus curves. J Cataract Refract Surg. 2013;39:873-880.
6. Lapid-Gortzak R, Bala C, Schwiegerling J, Suryakumar, BSOptom R. A novel methodology for measuring intraocular lens performance via acuity reserve. J Cataract Refract Surg. December 18, 2020. doi: 10.1097/j.jcrs.0000000000000561
7. Fernández J, Rodríguez-Vallejo M, Martínez J. Bridging the gap between lab and clinical practice in assessing multifocal IOLs. Cataract & Refractive Surgery Today Europe. October 2016. Accessed January 27, 2021. http://crstodayeurope.com/articles/2016-febbridging-the-gap-between-lab-and-clinical-practice-inassessing-multifocal-iols/
8. Fernández J, Rodríguez-Vallejo M, Tauste A, Albarrán C, Basterra I, Piñero D. Fast measure of visual acuity and contrast sensitivity defocus curves with an iPad application. Open Ophthalmol J. 2019;13:15-22.
9. Fernández J, Rodríguez-Vallejo M, Martínez J, Burguera N, Piñero DP. Pupil dependence assessment with multifocal intraocular lenses through visual acuity and contrast sensitivity defocus curves. Eur J Ophthalmol. July 3, 2020. doi:10.1177/1120672120940202
10. Fernández J, Rodríguez-Vallejo M, Martínez J, Burguera N, Piñero DP. Prediction of visual acuity and contrast sensitivity from optical simulations with multifocal intraocular lenses. J Refract Surg. 2019;35:789-796.
11. Fernández J, Rodríguez-Vallejo M, Martínez J, Tauste A, Piñero DP. Patient selection to optimize near vision performance with a low-addition trifocal lens. J Optom. 2020;13:50-58.
12. Fernández J, Rodríguez-Vallejo M, Martínez J, Burguera N, Piñero D. New system to measure chromatic contrast sensitivity defocus curves with multifocal intraocular lenses. ESCRS. Accessed January 27, 2021. https://www.escrs.org/amsterdam2020/programme/posters-details.asp?id=36495



