

In 2014, more than 8,600 corneas recovered by US eye banks were placed in long-term storage.1 Many of these corneas could have been recovered, screened, released, and used for intermediate transplantation (ie, within 2 weeks of tissue recovery) but were not because of low endothelial cell counts and/or a lack of surgical need. Instead, some corneas were preserved using limited long-term storage options such as glycerol and alcohol preservation. Tissues preserved by these methods, however, are not rendered sterile and require extensive reconstitution before use, lengthening operating procedure times while surgeons wait for the tissue to become amenable to surgical use.
Another 23,230 corneas were used for research or training.1 It is unknown how many of these corneas were medically suitable for transplantation but were unused because of a lack of long-term storage options at the recovering eye banks.
It is possible that updated methods for long-term corneal preservation would improve the use of donor corneas suitable for transplantation and expand the pool of corneal tissue available for ophthalmic procedures beyond traditional corneal transplantation. This idea combined with a desire to maximize the use of tissue that is medically suitable for transplantation led Lions VisionGift (LVG) to pursue an alternative method for preserving corneal tissue for long-term use. The goals of this effort are outlined below in the sidebar.
Goals for Preserving Corneal Tissue for Long-Term Use
- To extend the usable shelf-life for corneal tissue
- To store the corneal tissue at room temperature
- To eliminate the necessity of reconstitution and rehydration
- To be able to handle the tissue in the same manner as fresh corneal tissue with similar or stronger biomechanical properties
- To improve overall safety through tissue sterilization
ADVANTAGES OF E-BEAM IRRADIATION
LVG developed a patented method to preserve corneal tissue by electron-beam (e-beam) irradiation. LVG is an FDA-registered organization accredited by the Eye Bank Association of America, so it was imperative that the preservation process meet the medical standards of these agencies.
There are several advantages to using e-beam–irradiated corneal grafts instead of fresh tissue for lamellar applications.
No. 1: The availability of grafts that can be stored for up to 2 years at room temperature (2–38 °C) eliminates obstacles to the procurement of tissue for specific procedures.2,3 The graft is stored in recombinant human serum albumin, and the corneal tissue is ready for use straight from its sterile packaging. Unlike grafts preserved in glycerin or alcohol, it does not need to be reconstituted.
No. 2: The use of irradiated corneal grafts minimizes the risk of bacterial and fungal infections from contaminated donor tissue. The reason is that irradiated grafts are sterilized to a sterility assurance level of 10-6.
No. 3: Laboratory studies have shown that the clarity of irradiated, full-thickness corneas is similar to that of fresh corneas.3,4 This allows the former to be used in patch graft and lamellar applications over the visual axis without impeding the patient’s vision.
APPLICATIONS
Tissue sterility, an extended shelf-life, and greater availability make e-beam–irradiated corneas such as LVG's halo grafts an ideal alternative to fresh corneal tissue for numerous ophthalmic applications when a viable endothelium is not required. These corneal grafts have been used to cover glaucoma drainage tubes with excellent long-term outcomes (Figure 1).2 They have also been employed as patch grafts to treat corneal melts and corneal lesions (Figure 2).
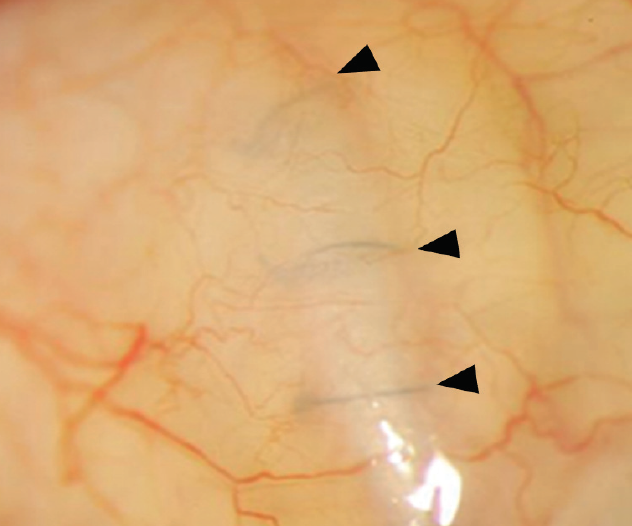
Figure 1. A graft was used to cover a glaucoma drainage device. The clear cornea allows surgeons to visualize the sutures (arrowheads) covered by the graft.
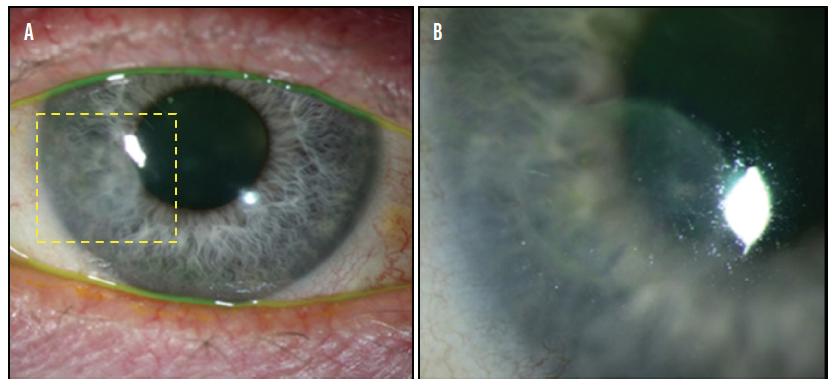
Figure 2. A lamellar graft was used to treat a corneal melt. This image was taken 1 year after surgery (A). The outlined area is magnified in B.
Figures 1 and 2 reprinted with permission from Springer Nature3
Recent advances in technology and surgical technique have made it possible to use irradiated grafts during refractive procedures such as the placement of corneal inlays (Figure 3) and intrastromal corneal ring segments for the treatment of keratoconus (Figure 4).
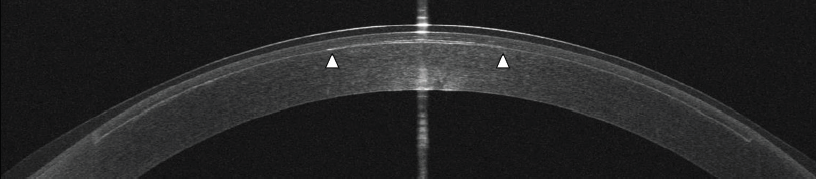
Figure 3. An OCT scan was obtained 10 minutes after the implantation of a lenticule in the anterior cornea for presbyopia correction. This lenticule is 20 µm thick (1/5 the thickness of a human hair) and is 2.5 mm in diameter (edges indicated by white arrowheads). The LASIK flap interface can also be seen.
Courtesy of Allotex
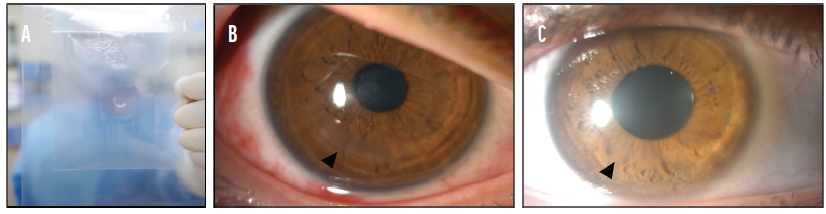
Figure 4. A sterile intrastromal corneal ring segment (ICRS) was stored in recombinant human serum albumin. A patented double-pouch design makes it easy to locate the tissue in the container while maintaining sterility in the OR (A). An ICRS was implanted to treat keratoconus. The eye is shown 1 month (B) and 6 months (C) after implantation of the ICRS.
B and C courtesy of Aylin Kiliç, MD
E-beam–sterilized corneas are expanding the possibilities of ophthalmic surgery.
1. Eye Bank Association of America. 2014 Eye Banking Statistical Report. Eye Bank Association of America; 2015. Accessed January 21, 2021. https://restoresight.org/wp-content/uploads/2015/03/2014_Statistical_Report-FINAL.pdf
2. Passo RM, Hoskins ZB, Tran KD, et al. Electron beam irradiated corneal versus gamma-irradiated scleral patch graft erosion rates in glaucoma drainage device surgery. Ophthalmol Ther. 2019;8(3):421-426.
3. Tran KD, Li Y, Holiman JD, et al. Light scattering measurements in electron-beam sterilized corneas stored in recombinant human serum albumin. Cell Tissue Bank. 2018;19(1):19-25.
4. Chae JJ, Choi JS, Lee JD, et al. Physical and biological characterization of the gamma-irradiated human cornea. Cornea. 2015;34(10):1287-1294.



