CASE PRESENTATION
A 32-year-old man seeks options for maximizing his spectacle independence. On presentation, BCVA is -3.75 -1.75 x 3º = 20/20 OD and -2.75 -0.50 x 170º = 20/40 OS.
An examination finds the quality of the ocular surface to be good. A relatively faint scar is evident in the anterior third of the left cornea starting at the 2-mm zone and extending to the temporal cornea at the 5-mm zone. Under mesopic conditions, the diameter of the pupil in each eye is 6.5 mm. Otherwise, the examination is unremarkable (Figures 1–3).
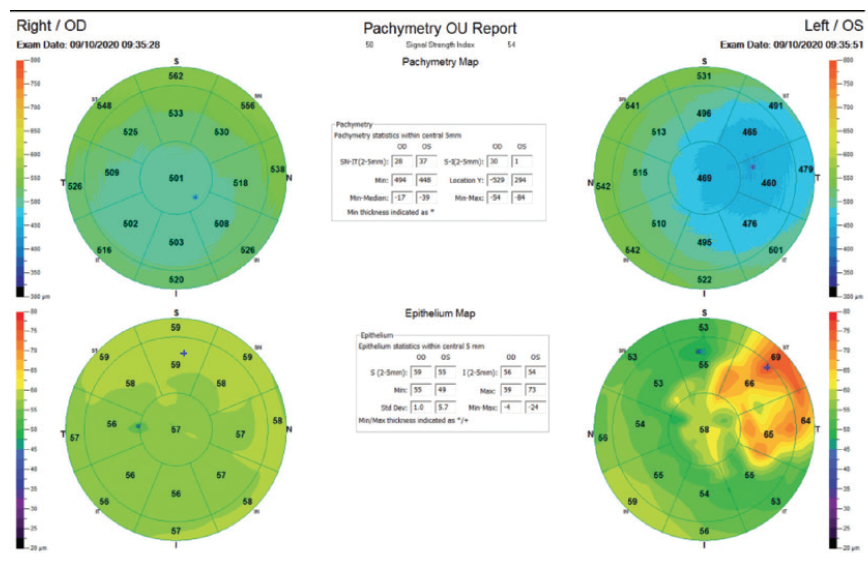
Figure 1. OCT epithelial thickness maps for the right and left eyes.
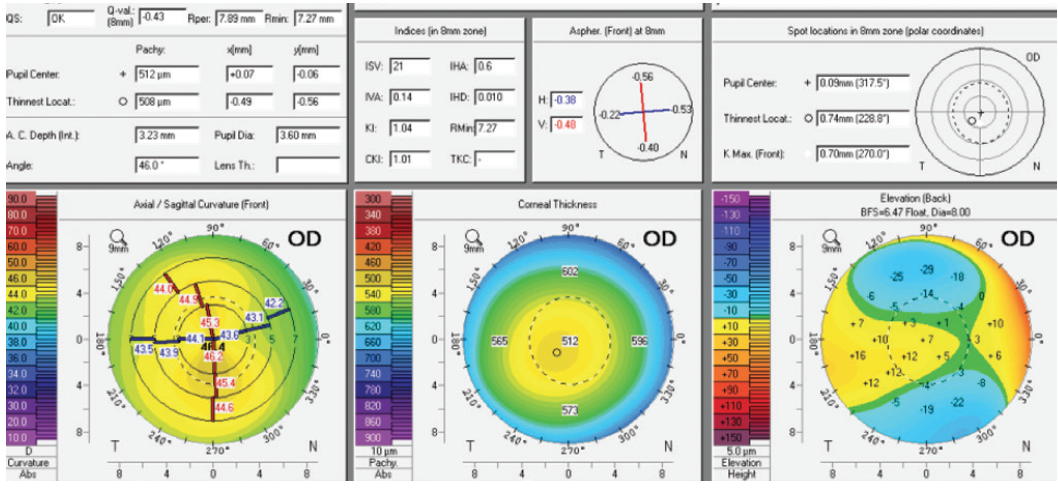
Figure 2. Measurements of the right eye obtained with the Pentacam (Oculus Optikgeräte).
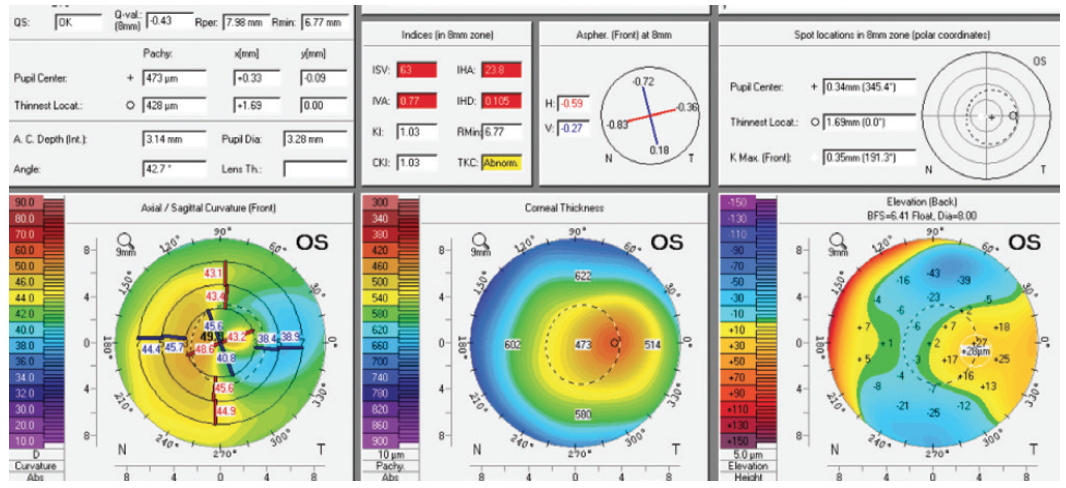
Figure 3. Measurements of the left eye obtained with the Pentacam.
The patient states that he does not wish to wear contact lenses because of psychological trauma from an experience with severe microbial keratitis secondary to contact lens wear in the left eye 4 years ago. How would you proceed?
—Case prepared by Allon Barsam, MD, MA, FRCOphth

ROMESH ANGUNAWELA, BM, MD, FRCOPHTH, FRCS, CERT LRS
This interesting case reflects a common scenario after a contact lens–related infection. In this situation, I routinely perform epithelial thickness mapping with OCT (Optovue or CSO MS-39) to get a better idea of how the differential epithelial and stromal thicknesses relate to the overall topography of the cornea. The scar in this patient’s left eye has created a temporal area of flattening with compensatory epithelial hyperplasia. This irregular shape reduces BCVA.
The epithelium acts as a natural masking mechanism that compensates for corneal irregularity. I would attempt to improve the overall shape of the cornea by performing transepithelial phototherapeutic keratectomy (PTK), using a setting that is 15 to 20 µm greater than the maximal epithelial thickness (in this case 86 µm) for an 8.5-mm treatment zone. Once the ablation depth reaches maximal epithelial thickness, I would stop the ablation and moisten the cornea with a moderately wet sponge to achieve fluid masking before completing the ablation. Because of the previous scarring and the propensity for haze formation, I would apply mitomycin C to the stromal bed for 40 to 50 seconds.
I would delay refractive correction of this eye for 6 to 12 months after PTK, at which time I would assess BCVA and the refraction. Additionally, epithelial thickness mapping would be repeated, and I would take this information into account when planning the refractive surgery procedure. Transepithelial PRK using the Schwind Amaris 1050RS laser (Schwind eye-tech-solutions) would be my preference.
For the right eye, my preference would be LASIK. Keratometry readings obtained with the Pentacam support the safety of this option in this case.
The preoperative discussion would be geared toward tempering the patient’s expectations. Informed consent would include an explanation that treating the left eye will require multiple steps and emphasize the possible need for a postoperative refractive enhancement. The patient should also understand that vision in the right eye may always be clearer than in the left eye but that binocular vision should be very good postoperatively.

STEPHEN C. COLEMAN, MD
The complexity of this case cannot be overstated. The primary issue is the decreased BCVA in the left eye and how to address it. Presumably, because the scar is described as faint and it does not extend fully into the visual axis, the decreased BCVA is due to irregular astigmatism and not the opaqueness or density of the scar. A slit-lamp photograph and a refraction over a rigid contact lens would help to determine the best approach to take.
It is important to confirm that the scar was indeed caused by a microbial pathogen and is not herpetic in nature, because herpes can sometimes be reactivated by the wavelength of the excimer laser. The overall thin pachymetry of the left eye precludes LASIK even if an extremely thin flap is created. An intraocular procedure could address the refractive error, but it would not improve BCVA. That leaves PRK.
The pachymetry map reveals that the corneal scar is associated with a significant loss of stromal tissue, and the epithelial map shows that the epithelium has done a good job of compensating. The hypertrophic epithelium over the scar has worked so well, in fact, that it has resulted in what is described as a “good” ocular surface. This is impressive.
At a minimum, a staged or stepwise approach may be offered to this patient. The first step is to normalize the corneal surface either with a topography-guided system or by using a blocking agent. Postoperatively, the overlying epithelium will presumably grow back uniformly, and then a refractive PRK may be performed. Without first normalizing the underlying corneal shape, the epithelium will regrow in an irregular, hypertrophic manner.
A conservative approach would be to perform LASIK on the right eye, ideally resulting in 20/20 UCVA, and a two-step PRK procedure on the left eye with the expectation of achieving 20/40 UCVA. A better outcome in the left eye would be considered a bonus. If the right eye is dominant, this strategy may satisfy the patient’s stated goal of maximizing his spectacle independence.

TOBIAS H. NEUHANN, MD, FEBOS-CR
Topography and tomography reveal slight asymmetrical astigmatism in the right eye. Epithelial mapping shows discrete thinning in the lower part of the cornea. Because the indices on the Pentacam do not suggest pathologic abnormality, forme fruste keratoconus need not be considered at present. The topographic findings are suspicious at best given the elevation and epithelial maps. Because the right eye is the patient’s better-seeing eye, I would avoid corneal refractive surgery.
Instead, I would recommend the placement of an Evo Visian Toric ICL (STAAR Surgical). The anterior chamber depth is 3.23 mm. A -3.50 D IOL powered to correct 2.00 D of astigmatism has an optic diameter of 5.8 mm, so the effective optical zone at the corneal plane measures 7.31 mm. This will compensate amply for the 6.5-mm mesopic pupil. Negative dysphotopsia is therefore not to be expected, although the central opening of the phakic IOL may initially cause nocturnal dysphotopsia. In my experience, patients cease to notice this phenomenon 3 to 12 months after surgery. If corneal topography changes in the future from forme fruste keratoconus, CXL may be performed despite the presence of an ICL—a major advantage of this strategy.
Based on the corneal abnormalities, discrete stromal opacity, and pachymetry measurements, I would not offer corneal refractive surgery for the left eye, either. My preference would be to implant a spherical Evo Visian ICL. In similar situations, I have found this procedure to improve visual acuity by 1 line. The appeal of ICL implantation in this eye is threefold. First, the slightly abnormal corneal topography can be monitored while left untreated. Second, the IOL may be removed if the patient does not tolerate the correction. Third, if a cataract forms, say in 40 years, the IOL power calculation will be easier to perform than after corneal refractive surgery.

ALEKSANDAR STOJANOVIC, MD, PHD
Laser vision correction in the right eye (-3.75 D sphere and -1.75 D cylinder) with a 6.5-mm optical zone would likely consume approximately 80 µm of stroma centrally. LASIK with a 110-µm flap would leave around 310 µm of residual stroma. The Pentacam measurements show that the thinnest portion of the cornea is close to where the maximum keratometry reading was obtained on the anterior surface, where the posterior elevation is also increased. Nevertheless, the epithelial thickness map shows an almost normal distribution. Surface ablation would be a safer option than LASIK, but both approaches should be okay. Alternatively, an Evo Visian ICL may be considered.
In the left eye, although the thinnest pachymetry reading corresponds to the area of highest posterior elevation, an ectatic process may be excluded because epithelial mapping at the same position shows thickening and the anterior curvature is flattened. The epithelium has filled in for the stromal tissue lost to keratitis. LASIK is contraindicated in this eye. Transepithelial topography-guided PRK would be my preference. (If that option is not available, topography-guided PRK can be combined with transepithelial PTK.) Transepithelial topography-guided PRK would address the relatively low refractive error as well as the surface irregularity and the superficial corneal opacity, which are the most probable causes of the reduced corrected distance visual acuity. Assuming that the stromal ablation depth does not exceed 55 µm in addition to the epithelial ablation of 60 µm, the treatment will leave a residual stromal thickness of 345 µm centrally. The implantation of an ICL would not address the corneal optical irregularity or the stromal opacity.

WHAT I DID: ALLON BARSAM, MD, MA, FRCOPHTH
The right eye underwent LASIK using a 6.3-mm optical zone and a 100-µm flap created with a femtosecond laser. The left eye underwent transepithelial topography-guided PRK with mitomycin C. Three months after surgery, UCVA was 20/16 OD and 20/25 OS.



